About CancerPeutics
- CancerPeutics is working on a project to develop a CAR-T therapeutics that is currently in the spotlight in the field of cancer treatment. The company is conducting a joint study with the KRICT (Korea Research Institute of Chemical Technology) and KRIBB (Korea Research Institute of Bioscience and Biotechnology) on the development of the CAR-T therapeutics. The researchers in the institutions, who work on the CAR-T and CAR-NK fields are well known to have the best levels in the country and research fields. The company will have an advanced infrastructure and technical skills in developing CAR-T treatments through a synergy with foreign companies and institutions related to CAR-T and domestic institutions.
- To aim at the global license out, CancerPeutics has hired Peter Hollands, a professor of global immuno-cell therapy, as a CTO since the beginning of CancerPeutics’ establishment and has been developing a CAR-T treatment in the line of the global standard. In line with Professor Peter's development command, the KRICT and the KRIBB has signed an agreement on technology introduction and joint research.
- Dr. Chi-Hoon Park from the KRICT will lead the development of the new CAR-T treatment; based on the patents applied. Also, Dr. Ji-Yoon Noh from the KRIBB will develop the CAR-T treatment as a scientist who led the development of the CAR-NK in the CiM (Customized i-Medicine).
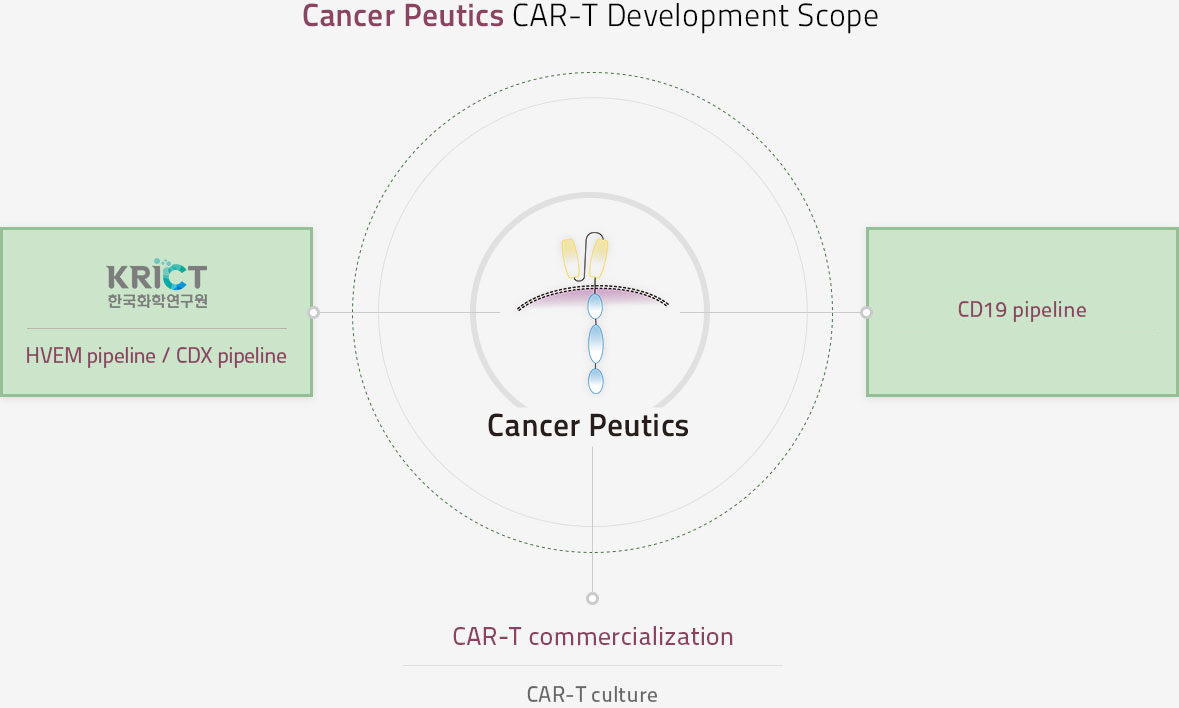
- Three pipelines were formed through this joint study. GCT-201 is the commercialization of HVEM targeted CAR-T treatment. Because HVEM is mainly expressed in melanoma, HVEM targeted CAR-T can be used as a treatment for melanoma. It aims to test its nonclinical trial in 2020. GCT-301 is Novel CAR-T development about a new target, which discovered by Chi-Hoon Park, a researcher at the KRICT. As soon as the design for the development of Novel CAR-T is completed, the company plans to begin research for clinical trials so that the second CAR-T treatment can be released immediately after the first CAR-T treatment. GCT-401 is developing a CD19 target CAR-T
The reason for the research
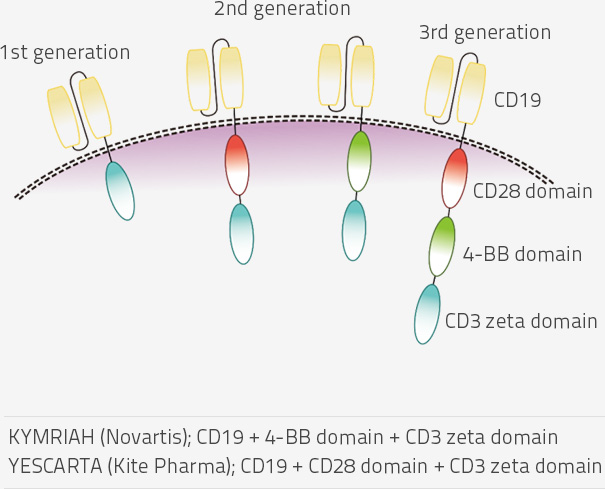
- KYMRIAH (Novartis) and YESCARTA (Kite Pharma) are approved by FDA for leukemia, and both are second-generation CAR-T therapeutics.
- Research on fourth-generation CAR-T has better therapeutic effects without a side effect.
- CancerPeutics conduct researching on fourth-generation CAR-T therapeutics.

